Neuro Device Directory
Explore FDA-cleared neurological medical devices with our curated, high-fidelity directory.
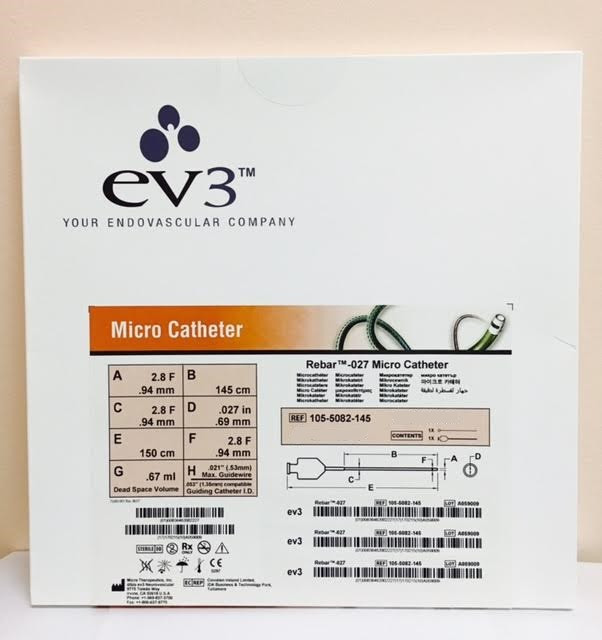
Plato MICROCATH® 27 Microcatheter
The Plato MICROCATH® 27 Microcatheter is intended to assist in the delivery of diagnostic and/or therapeutic agents to the peripheral and neurovascular systems.
PROTÉGÉ Carotid Stent System
The PROTÉGÉ GPS and RX Carotid Stent Systems are intended for the endovascular treatment of carotid artery atherosclerotic disease to open partially blocked arteries, hold plaque against the artery wa

ENVOY® Distal Access (DA) Guiding Catheter
The ENVOY Distal Access (DA) Guiding Catheter is intended for use in the peripheral, coronary, and neuro vasculature for the intravascular introduction of interventional/diagnostic devices.

ASAHI Tellus
This microcatheter is a medical device intended for angiography and/or infusion of various substances including diagnosis, embolization and treatment in the peripheral vasculature using an appropriate
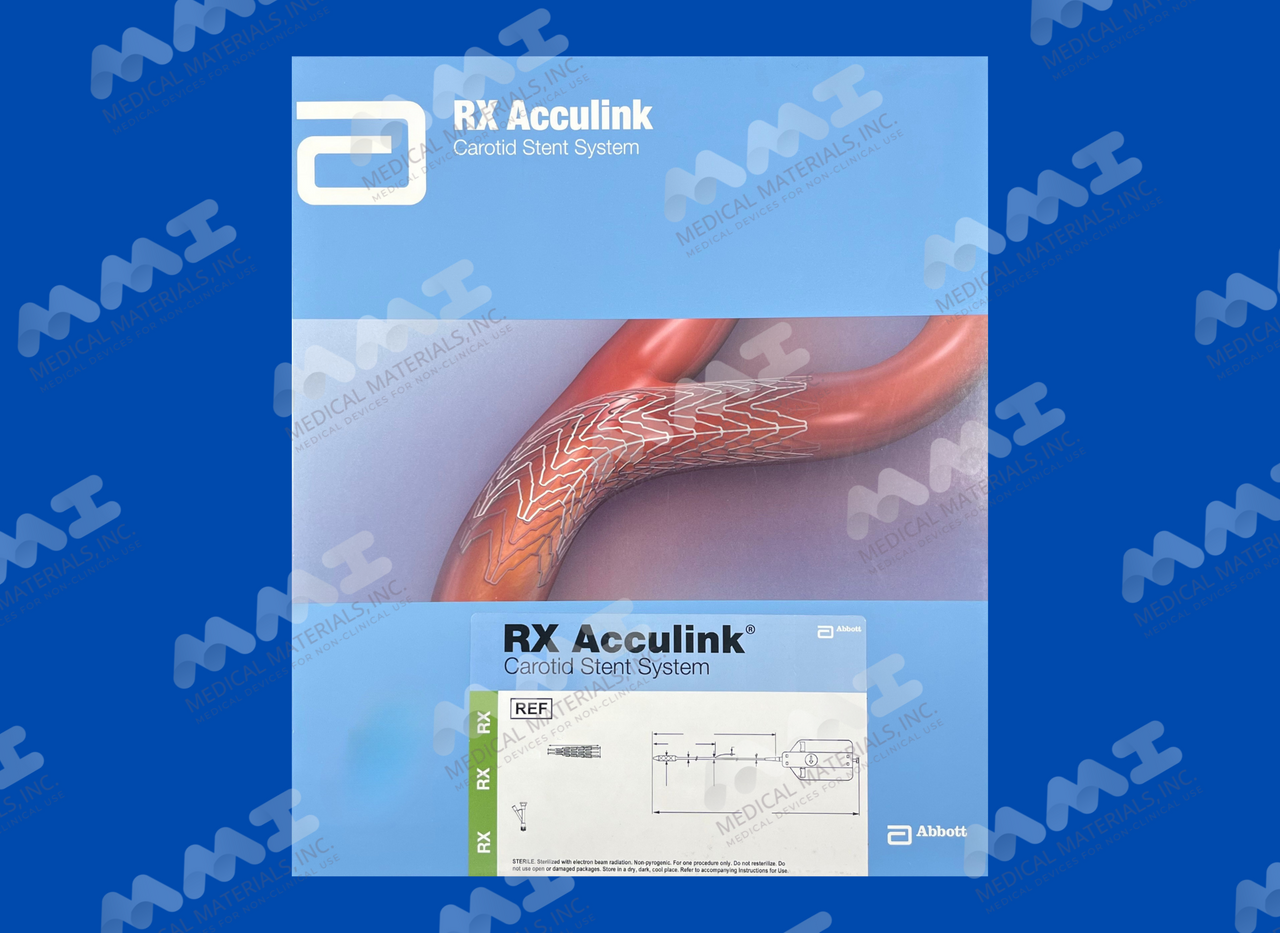
RX Acculink Carotid Stent System
The RX Acculink Carotid Stent System, used in conjunction with the Abbott Vascular embolic protection system specified below, is indicated for the treatment of patients at high and standard risk for a
Alto™ Abdominal Stent Graft System
The Alto™ Abdominal Stent Graft System is indicated for treatment of patients with infrarenal abdominal aortic aneurysms having the vascular morphology suitable for endovascular repair with the device
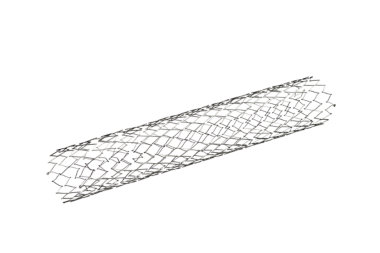
Artificial Embolization Device

Micrus® Ascent® Occlusion Balloon Catheter
The Micrus Ascent Occlusion Balloon Catheters are intended for use in the blood vessels of the peripheral and neuro vasculature where temporary occlusion is desired and offers a vessel selective techn
Benchmark BMX96 Access System
The Benchmark BMX96 Access System is indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature.

072 Aspiration System
072 Aspiration Catheter: As part of the 072 Aspiration System, the 072 Aspiration Catheter with a compatible suction pump is indicated for use in the revascularization of patients with acute ischemic

Axium™ Detachable Coil
Axium™ Detachable Coil: Axium™ Detachable Coils are intended for the endovascular embolization of intracranial aneurysms. Axium™ Detachable Coils are also intended for the embolization of other neuro
Barricade Embolization Coil System
The Barricade Coil System is intended for the endovascular embolization of Intracranial aneurysm and other neurovascular abnormalities such as arteriovenous malformations and arteriovenous fistulae. T
Bead Block
Bead Block microspheres are intended to be used for the embolization of hypervascular tumours, including uterine fibroids (UFE) and arteriovenous malformations (AVMs). Bead Block microspheres are also
CEREGLIDE™ 42 Intermediate Catheter; CEREGLIDE™ 57 Intermediate Catheter
The CEREGLIDE 42 Intermediate Catheter and the CEREGLIDE 57 Intermediate Catheter, with the Cerenovus Aspiration Tubing Set and a compatible aspiration pump, are indicated for use in the revasculariza

Imager 11 Angiographic Catheter
The Imager 11 Angiographic Catheters are designed to provide a pathway for delivering contrast media to selected sites in the vascular system including the carotid arteries. Additionally, the 5F Selec
CalliSpheres Embolic Microspheres
Callispheres Embolic Microspheres and 8Spheres Embolic Microspheres are intended to be used for the embolization of arteriovenous malformations (AVMs) and hypervascular tumors, including uterine fibro
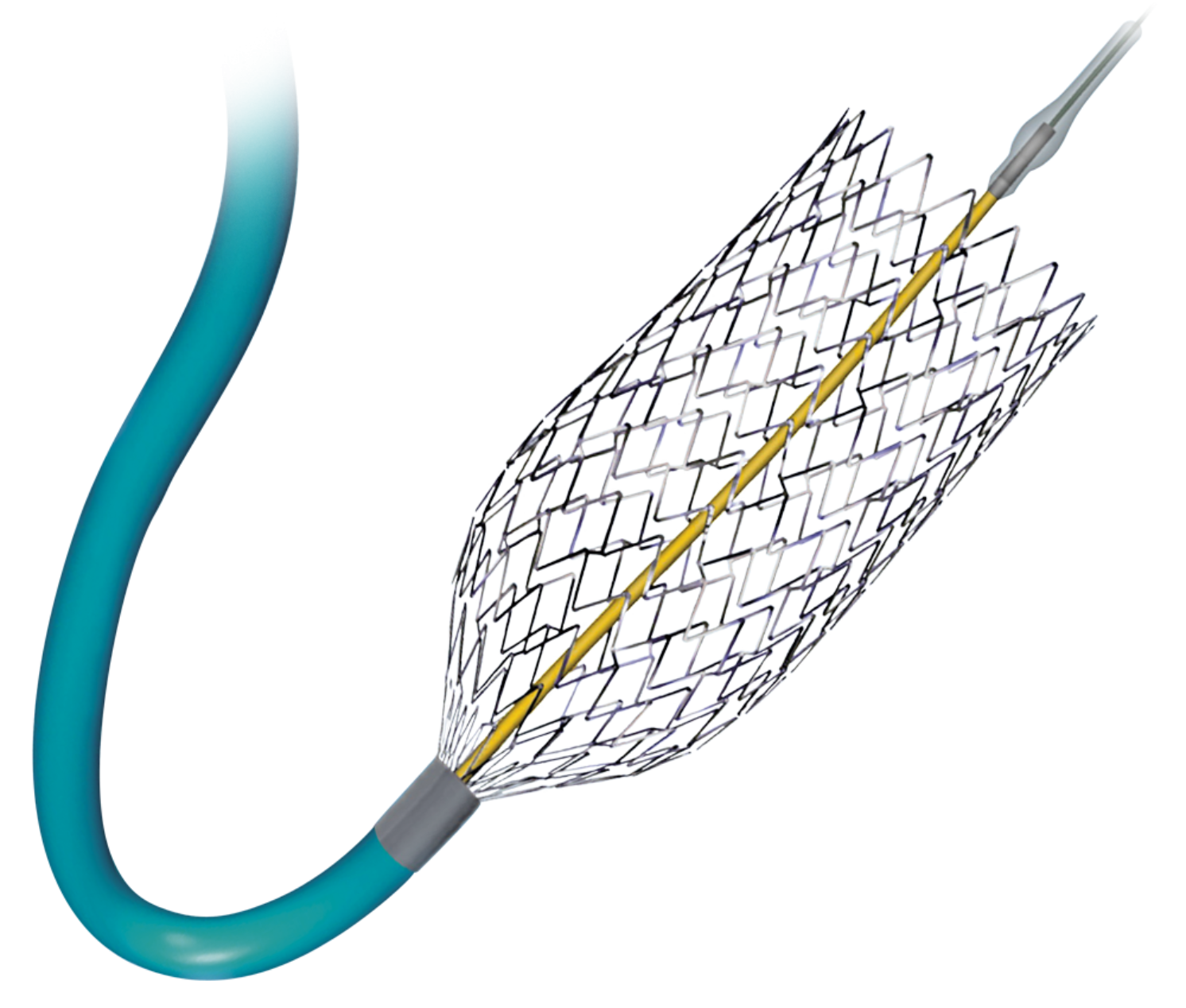
Cordis PRECISE Nitinol Stent System
The Cordis PRECISE Nitinol Stent System used in conjunction with the ANGIOGUARD™ XP Emboli Capture Guidewire is indicated for the treatment of patients at high risk for adverse events from carotid end

Carotid WALLSTENT Monorail Endoprosthesis
The Carotid WALLSTENT Monorail Endoprosthesis (Carotid WALLSTENT Endoprosthesis), used in conjunction with the Boston Scientific embolic protection system, is indicated for the treatment of patients a

Contour SE™ Microspheres
Contour SE™ Microspheres are indicated for use for the embolization of hypervascular tumors and arteriovenous malformations.
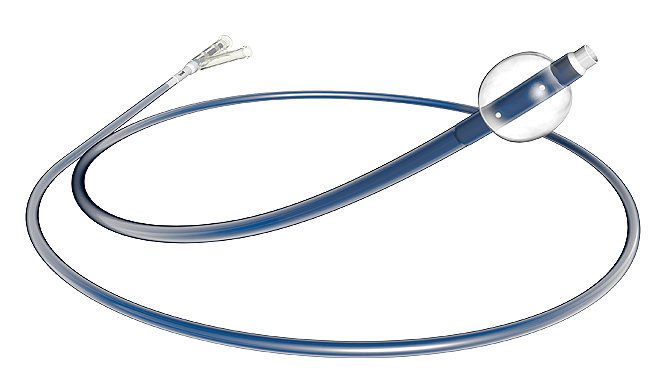
Concentric Balloon Guide Catheter
The Concentric Balloon Guide Catheter is indicated for use in facilitating and guidance of intravascular catheters into a selected blood vessel in the peripheral and neuro vascular systems. The balloo

Polyvinyl Alcohol Foam Embolization Particles
The Cook Polyvinyl Alcohol Foam Embolization Particles are intended for embolization of the blood supply to symptomatic uterine fibroids.
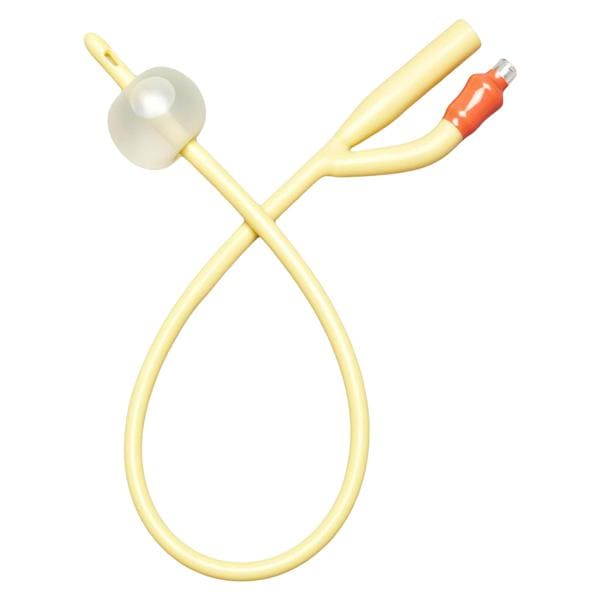
Slinky Catheter
The Slinky Catheter is indicated for general intravascular use, including the neuro and peripheral vasculature. It can be used to facilitate introduction of diagnostic agents and therapeutic devices.
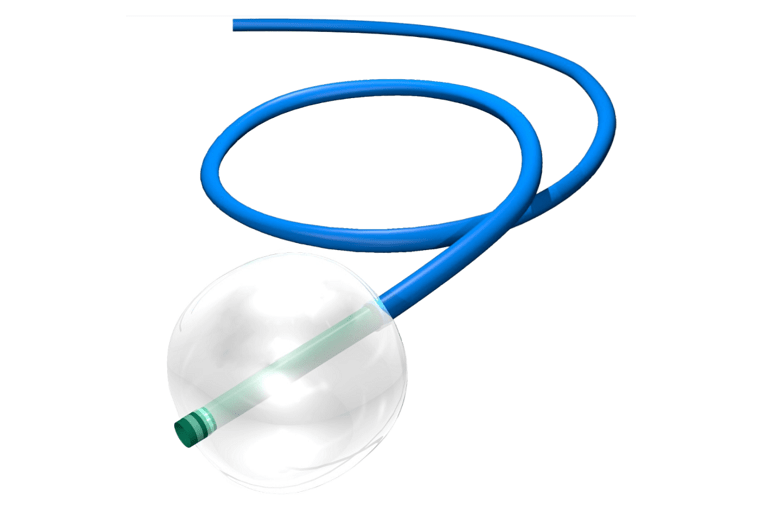
CELLO II Balloon Guide Catheter
The CELLO II Balloon Guide Catheter is indicated for use in facilitating the insertion and guidance of intravascular catheters into a selected blood vessel in the peripheral and neuro vasculature syst

MIVI Q Distal Access Catheter
The MIVI Q Distal Access Catheter is indicated for use with compatible guide catheters in facilitating the insertion and guidance of microcatheters into a selected blood vessel in the peripheral, coro
Sim&Size
Sim&Size enables visualization of cerebral blood vessels for preoperational planning and sizing for neurovascular interventions and surgery. Sim&Size also allows for the ability to computationally mod
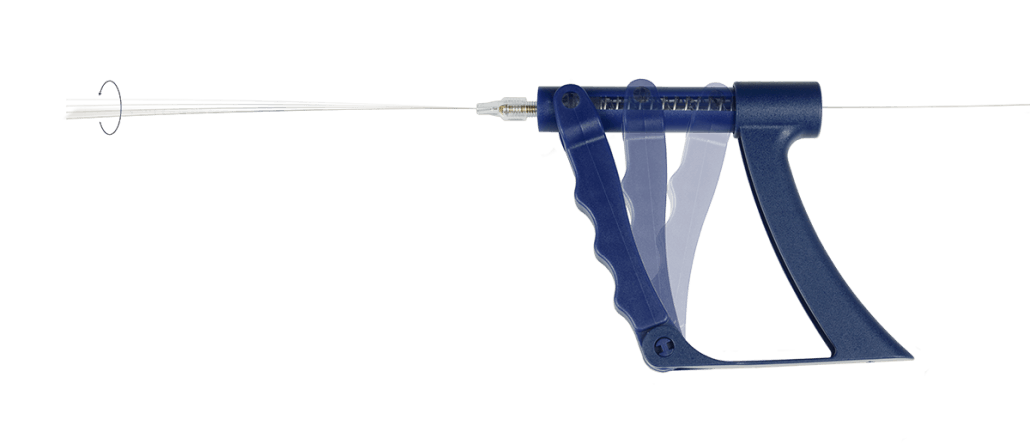
Distal Access Torque Device or Controller (Predict or Spinr)
Distal Access torque devices are used to maneuver guide wires in the coronary and peripheral vasculature during interventional or diagnostic procedures. Distal Access torque devices are not intended f

AXS Catalyst™ Distal Access Catheter
The AXS Catalyst Distal Access Catheter is indicated for use in facilitating the insertion and guidance of appropriately sized interventional devices into a selected blood vessel in the peripheral and

FUBUKI XF Neurovascular Long Sheath
This product is intended to be used to guide interventional devices for neurovascular therapy to a lesion or a procedural site for a percutaneous intravascular procedure in the neurovasculature. This
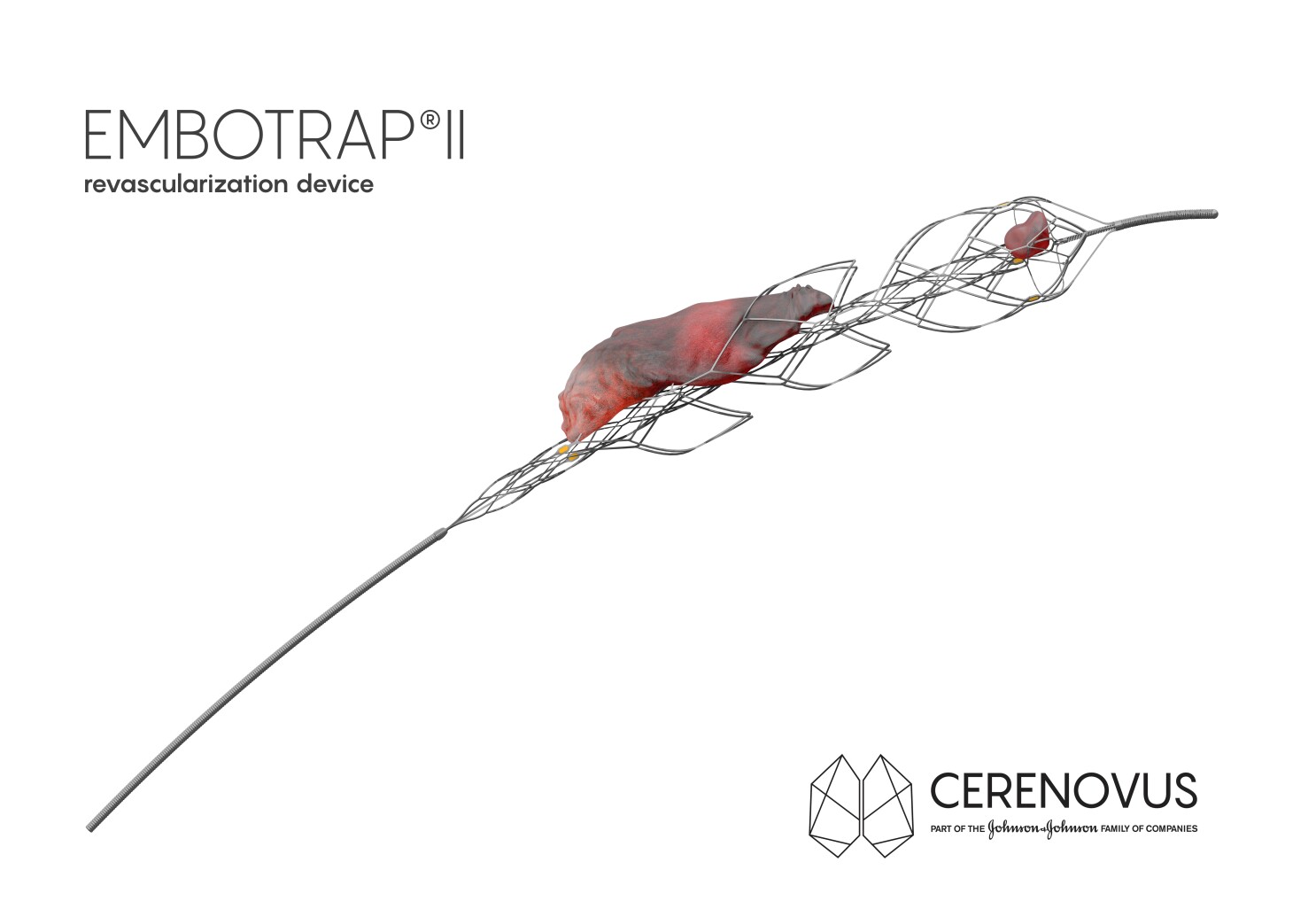
EmboTrap® II Revascularization Device
The EmboTrap® II Revascularization Device is intended to restore blood flow in the neurovasculature by removing thrombus in patients experiencing ischemic stroke within 8 hours of symptom onset. Patie

Excelsior XT-17 Microcatheter
Stryker Neurovascular's Excelsior XT -17 Microcatheters are intended to assist in the delivery of diagnostic agents, such as contrast media, and therapeutic agents, such as occlusion coils, into the p

NeVa VS
The NeVa VS is indicated for use as an adjunct treatment for symptomatic cerebral vasospasm in the internal carotid artery (ICA), middle cerebral artery (MCA), anterior cerebral artery (ACA), posterio
MicroVention Low-Profile Visualized Intraluminal Support (LVIS) and LVIS Jr.
The LVIS and LVIS Jr. are indicated for use with neurovascular embolization coils in patients ≥ 18 years of age for the treatment of wide-neck (neck width ≥ 4 mm or dome to neck ratio < 2) saccular in

Roadsaver™ Carotid Stent System
The Roadsaver Carotid Stent System, when used in conjunction with the Nanoparasol™ Embolic Protection System (EPS), is indicated for the treatment of carotid artery stenosis in patients with elevated

SolitaireTM FR Revascularization Device
The SolitaireTM FR Revascularization Device is intended to restore blood flow by removing thrombus from a large intracranial vessel in patients experiencing ischemic stroke within 8 hours of symptom o
Lava™ Liquid Embolic System
Lava™ LES is indicated for embolization of arterial hemorrhage in the peripheral vasculature.
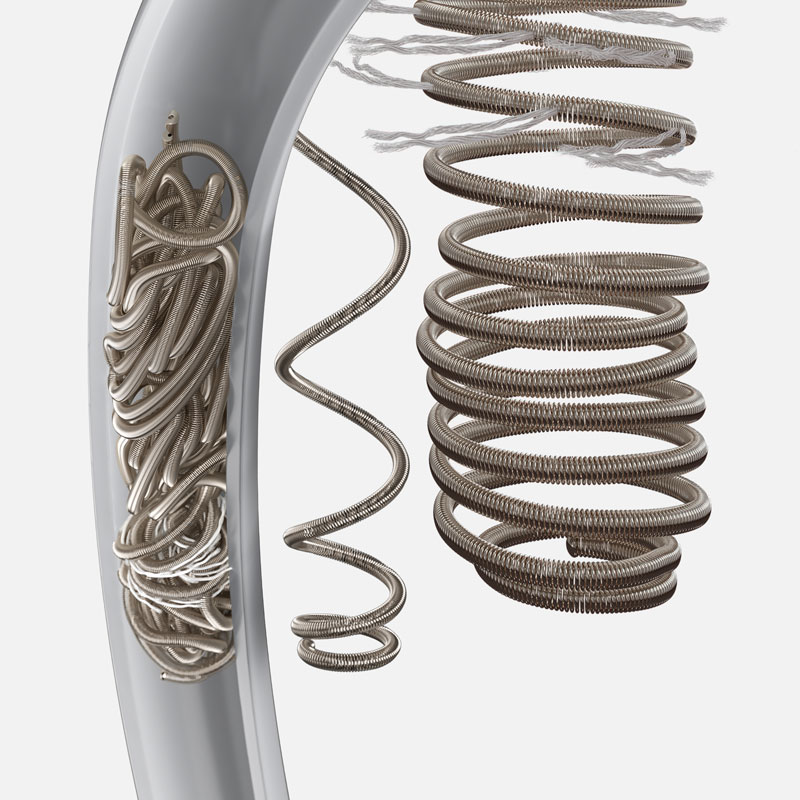
Sapphire NXT Detachable Coil System
The SapphireTM Detachable Coils are intended for the endovascular embolization of intracranial aneurysms that - because of their morphology, their location, or the patient's general medical condition
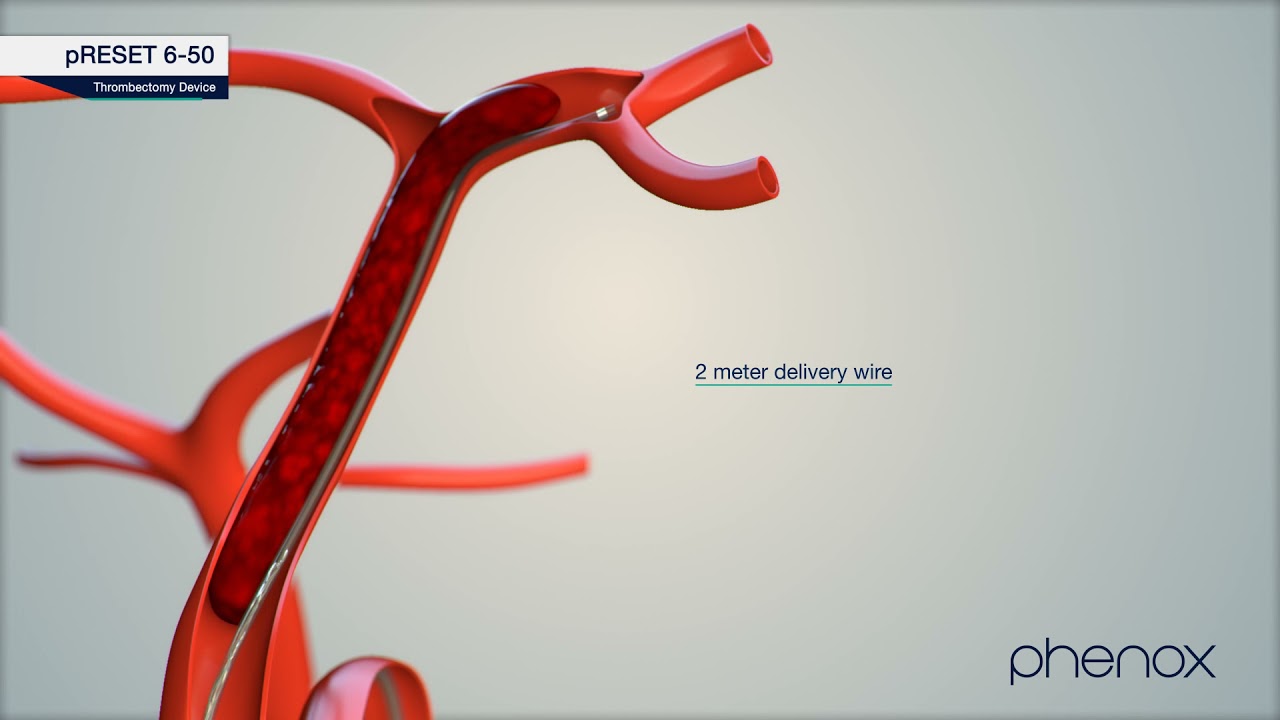
pRESET Thrombectomy Device
1. The pRESET® Thrombectomy Device is indicated for use to restore blood flow in the neurovasculature by removing thrombus for the treatment of acute ischemic stroke to reduce disability in patients w

Rist™ Radial Access Selective Catheter, Rist™ 071 Radial Access Guide Catheter, Rist™ 079 Radial Access Guide Catheter
The Rist™ 079 Radial Access Guide Catheter is indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature. The Rist™ 071 Radial Access Guide Catheter

Sim&Size
Sim&Size enables visualization of cerebral blood vessels for preoperational planning and sizing for neurovascular interventions and surgery. Sim&Size also allows for the ability to computationally mod

Sim&Size
Sim&Size enables visualization of cerebral blood vessels for preoperational planning and sizing for neurovascular interventions and surgery. Sim&Size also allows for the ability to computationally mod

Rist™ 071 Radial Access Guide Catheter
The Rist™ 071 Radial Access Guide Catheter is indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature.

FUBUKI XF-R Neurovascular Long Sheath
The FUBUKI XF-R Neurovascular Long Sheath is intended to be used to guide interventional devices for neurovascular therapy to a lesion or a procedural site for a percutaneous intravascular procedure i

Raptor Aspiration Catheter; Balt Aspiration Tubing Set
Raptor Aspiration Catheter: The Raptor Aspiration Catheter with a compatible aspiration pump and Balt Aspiration Tubing Set is indicated for use in the revascularization of patients with acute ischemi

SOFIA PLUS Distal Access Catheter
The SOFIA PLUS/Distal Access Catheters are indicated for general intravascular use, including the neuro and peripheral vasculature. It can be used to facilitate introduction of diagnostic and therapeu

EagleRay Long Sheath and EagleRay Access Catheter
The EagleRay Long Sheath and EagleRay Access Catheter are indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature.
087 Balloon Guide Catheter System
The 087 Balloon Guide Catheter System is indicated for use in facilitating the insertion and guidance of an intravascular catheter into a selected blood vessel in the peripheral and neuro vasculature.
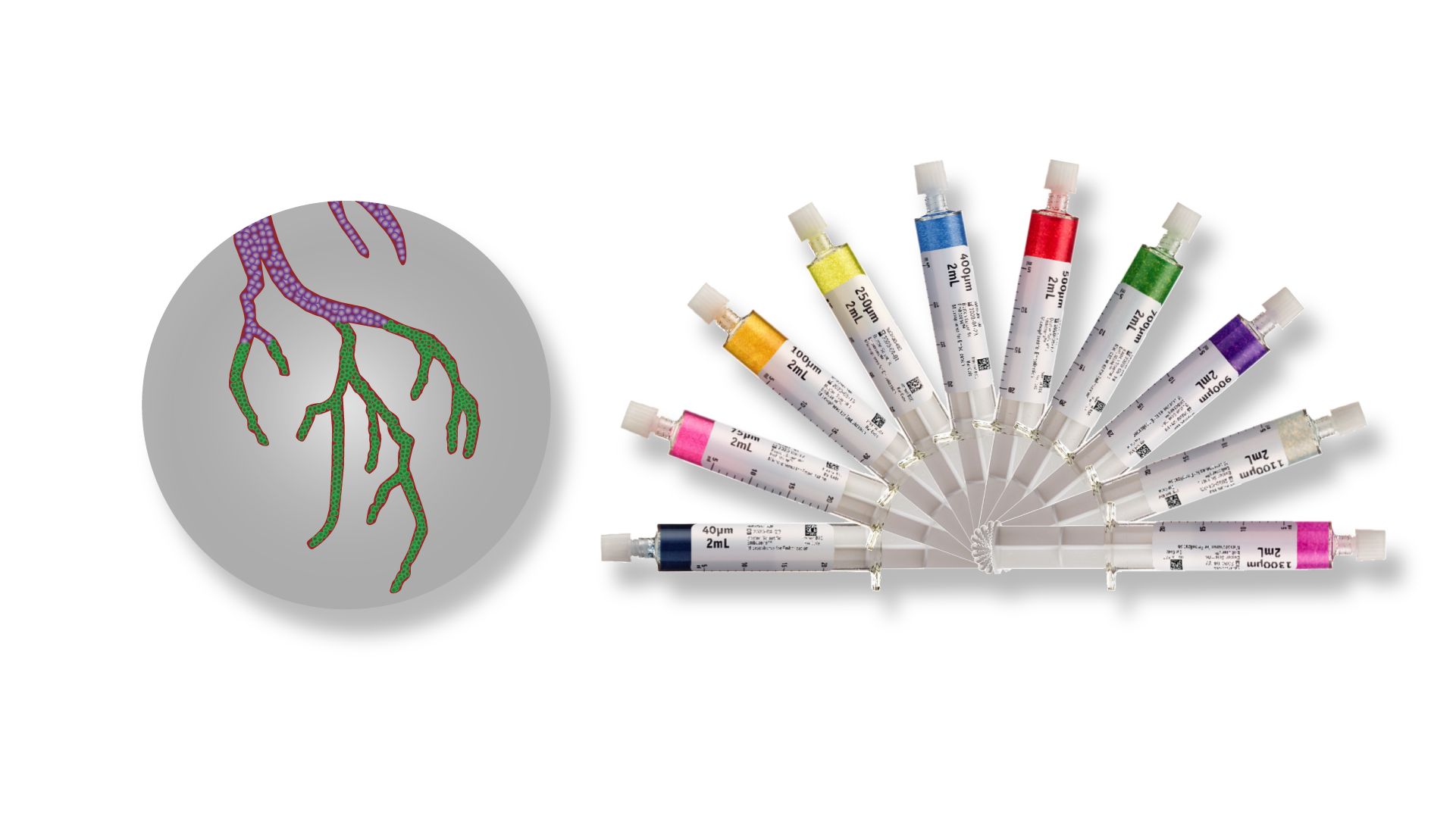
Embozene® Microspheres
Embozene® Microspheres are intended for embolization of arteriovenous malformations, and hypervascular tumors, including uterine fibroids.
Solitaire™ X Revascularization Device
1. The Solitaire™ X Revascularization Device is indicated for use to restore blood flow in the neurovasculature by removing thrombus for the treatment of acute ischemic stroke to reduce disability in
EMBOGUARD Balloon Guide Catheter
EMBOGUARD Balloon Guide Catheters are indicated for use in facilitating the insertion and guidance of an intravascular catheter into a selected blood vessel in the neurovascular system. The balloon pr
Benchmark™ Intracranial Access System
The Benchmark Intracranial Access System is indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature.
Tigertriever Revascularization Device
The Tigertriever Revascularization Device is intended to restore blood flow by removing thrombus from a large intracranial vessel in patients experiencing ischemic stroke within 8 hours of symptom ons
NG Delivery Catheter
The NG Delivery Catheter is intended for general intravascular use, including the peripheral, coronary and neuro vasculature for the infusion of diagnostic agents, such as contrast media, and to assis
EMBOGUARD Balloon Guide Catheter
EMBOGUARD Balloon Guide Catheters are indicated for use in facilitating the insertion and guidance of an intravascular catheter into a selected blood vessel in the neurovascular system. The balloon pr
Next Generation Aspiration Catheter; Balt Aspiration Tubing Set
[object Object]

Sim&Size
Sim&Size enables visualization of cerebral blood vessels for preoperational planning and sizing for neurovascular interventions and surgery. Sim&Size also allows for the ability to computationally mod
Optima Coil System
The Optima Coil System is intended for the endovascular embolization of intracranial aneurysms and other neurovascular abnormalities such as arteriovenous malformations and arteriovenous fistulae. The

Sapphire Detachable Fibered Coil System
Sapphire Detachable Coils (Non-Fiber): The Sapphire Detachable Coils are intended for the endovascular embolization of intracranial aneurysms that - because of their morphology, their location, or the

Bead Block™
Bead Block™ microspheres are intended to be used for the embolization of hypervascular tumors, including uterine fibroids and arteriovenous malformations (AVMs).

ERIC™ Retrieval Device
The ERIC Retrieval Device is indicated to restore blood flow in the neurovasculature by removing thrombus in patients experiencing ischemic stroke within 8 hours of symptom onset. Patients who are ine
MIDWAY Delivery Catheter (MIDWAY 43 Delivery Catheter; MIDWAY 62 Delivery Catheter)
The MIDWAY Delivery Catheter is indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature.

Tigertriever Revascularization Device
The Tigertriever Revascularization Device is intended to restore blood flow by removing thrombus from a large intracranial vessel in patients experiencing ischemic stroke within 8 hours of symptom ons
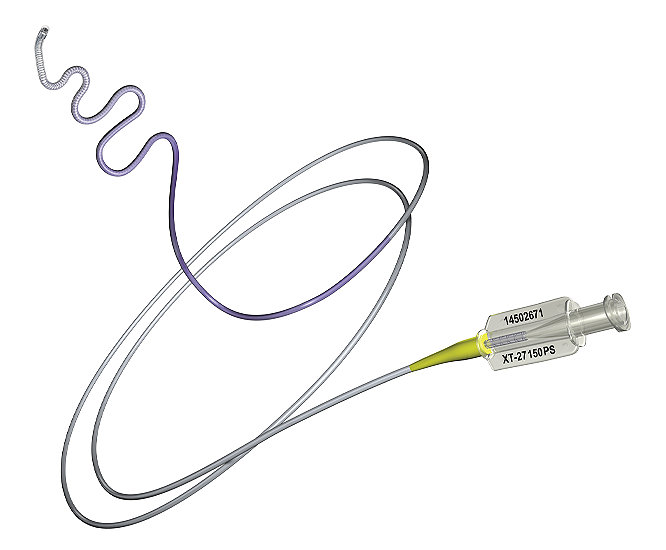
Excelsior XT-27 Microcatheter
The Excelsior XT-27 Microcatheter and Excelsior XT-27 Flex Microcatheter are intended to assist in the delivery of embolization particles, diagnostic agents (such as contrast media), and interventiona

6F Wallaby Long Sheath
The 6F Wallaby Long Sheath is indicated for the introduction of interventional devices into the peripheral and neuro vasculature.

Distal Access Catheter
The Distal Access Catheter is indicated for general intravascular use, including the neuro and peripheral vasculature. It can be used to facilitate introduction of diagnostic agents or therapeutic dev

Sunsphere
Sunsphere is intended to be used for the embolization of hypervascular tumors, including uterine fibroids, and arteriovenous malformations (AVMs).

Imperative Care Radial 088 Access System
The Imperative Care Radial 088 Access System is indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature.

RIST Radial Access Catheter
The RIST Radial Access Catheter is indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature.

BOSS™ Balloon Guide Catheter
The BOSS Balloon Guide Catheter is indicated for use in facilitating the insertion and guidance of an intravascular catheter into a selected blood vessel in the peripheral and neurovascular systems. T

Exponent Self-Expanding Carotid Stent with Over-the-Wire (OTW) Delivery System
The Medtronic Vascular Exponent Self-Expanding Carotid Stent with OTW Delivery System, used in conjunction with the Medtronic Vascular embolic protection system, is indicated for improving carotid lum
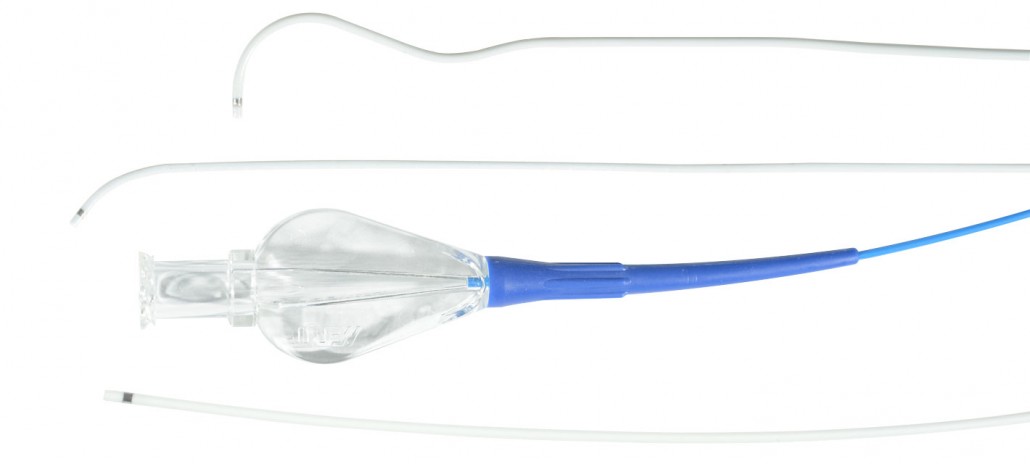
Maestro Microcatheter
The Microcatheter is intended for general intravascular use, including peripheral and coronary vasculature. Once the subselective region has been accessed, the microcatheter can be used for the contro
TrackerTM ExcelTM-14 Pre-Shaped Microcatheter
Boston Scientific's TrackerTM ExcelTM-14 Pre-Shaped Microcatheter is intended to assist in the delivery of diagnostic agents, such as contrast media, and therapeutic agents, such as occlusion coils in

Micrus® Ascent™ Occlusion Balloon Catheter and Micrus® Summit™ Occlusion Balloon Catheter
The Micrus® AscentTM and SummitTM Occlusion Balloon Catheters are intended for use in the blood vessels of the peripheral and neuro vasculature where temporary occlusion is desired and offers a vessel

Millipede 070 Aspiration Catheter
Millipede 070 Aspiration Catheter: The Millipede 070 Aspiration Catheter, with the Perfuze Aspiration Tube Set and a compatible aspiration pump, is indicated for use in the revascularization of patien

Micrus Courier 270 Microcatheter
Micrus Courier Microcatheters are intended to aid in the delivery of diagnostic agents, such as contrast media, as well as therapeutic agents, such as occlusion coils, into the peripheral, coronary an

Surpass Streamline Flow Diverter
The Surpass Streamline Flow Diverter is indicated for use in the endovascular treatment of patients (18 years of age and older) with unruptured large or giant saccular wide-neck (neck width ≥ 4 mm or

NexStent® Carotid Stent and Delivery System
The NexStent® System used in conjunction with FilterWire EZTM Embolic Protection System, is indicated for treatment of patients at high risk for adverse events from carotid endarterectomy (See Section
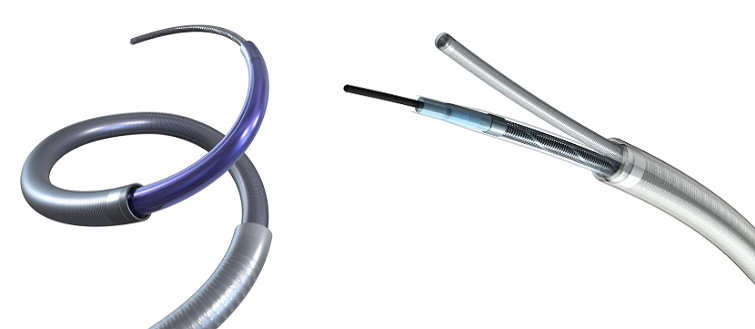
AXS Infinity LS Plus Long Sheath
The AXS Infinity LS Plus Long Sheath is indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature.

Trevo ProVue and XP ProVue Retrievers
The Trevo Retrievers are indicated for use to restore blood flow in the neurovasculature by removing thrombus for the treatment of acute ischemic stroke to reduce disability in patients with a persist

Numen Coil Embolization System
Numen Coil Embolization System is intended to endovascularly obstruct or occlude blood flow in vascular abnormalities of the neurovascular and peripheral vessels. Numen Coil Embolization System is ind

Concentric Balloon Guide Catheter
The Concentric Balloon Guide Catheter is indicated for use in facilitating the insertion and guidance of an intravascular catheter into a selected blood vessel in the peripheral and neuro vascular sys
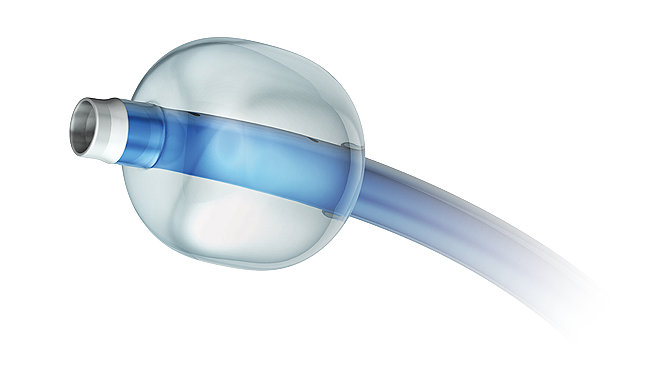
Modified FlowGate Balloon Guide Catheter
The Concentric Balloon Guide Catheter is indicated for use in facilitating the insertion and guidance of an intravascular catheter into a selected blood vessel in the peripheral and neuro vascular sys
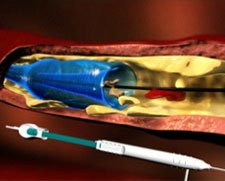
PROTEUS™ PTA Balloon Catheter with Embolic Capture Feature
The Angioslide PROTEUS™ Percutaneous Transluminal Angioplasty (PTA) Balloon Catheter with Embolic Capture Feature is indicated for peripheral transluminal angioplasty and for capture and containment o
CEREGLIDE™ 42 Intermediate Catheter; CEREGLIDE™ 57 Intermediate Catheter
The CEREGLIDE 42 Intermediate Catheter and the CEREGLIDE 57 Intermediate Catheter are indicated for use in facilitating the insertion and guidance of appropriately sized interventional devices into a

Paragon 8F Balloon Guide Catheter
The Paragon 8F Balloon Guide Catheter is indicated for use in facilitating the insertion and guidance of an intravascular catheter into a selected blood vessel in the peripheral and neuro vascular sys

Esperance 3+ Aspiration Catheter System
The Esperance 3+ Aspiration Catheter with the Wallaby Aspiration Tubing set and a compatible aspiration pump is intended for use in the revascularization of patients with acute ischemic stroke seconda

Onyx Liquid Embolic System
Presurgical embolization of brain arteriovenous malformations (bAVMs).

Onyx Liquid Embolic System Onyx HD-500
Onyx® Liquid Embolic System (Onyx® HD-500) is indicated for treatment of intracranial, saccular, sidewall aneurysms that present with a wide neck (> 4 mm) or with a dome-to-neck ratio < 2 that are not

Onyx Liquid Embolic System (Onyx LES)
The Onyx® Liquid Embolic System (hereinafter called the Onyx® LES) is indicated for the presurgical embolization of brain arteriovenous malformations (bAVMs).

Onyx Liquid Embolic System
The Onyx Liquid Embolic System is intended for pre-surgical embolization of brain arteriovenous malformations (AVMs) to reduce blood loss during surgical removal of the AVM.

PROWLER SELECT LP ES Microcatheter
The PROWLER SELECT LP ES Microcatheter is intended for the introduction of embolic devices and infusion of diagnostic agents into the peripheral and neuro vasculature. The PROWLER XS Microcatheter is

Gateway PTA Balloon Catheter
The Gateway PTA Balloon Catheter is indicated for balloon dilation of the stenotic portion of intracranial arteries prior to stenting for the purpose of improving intracranial perfusion.
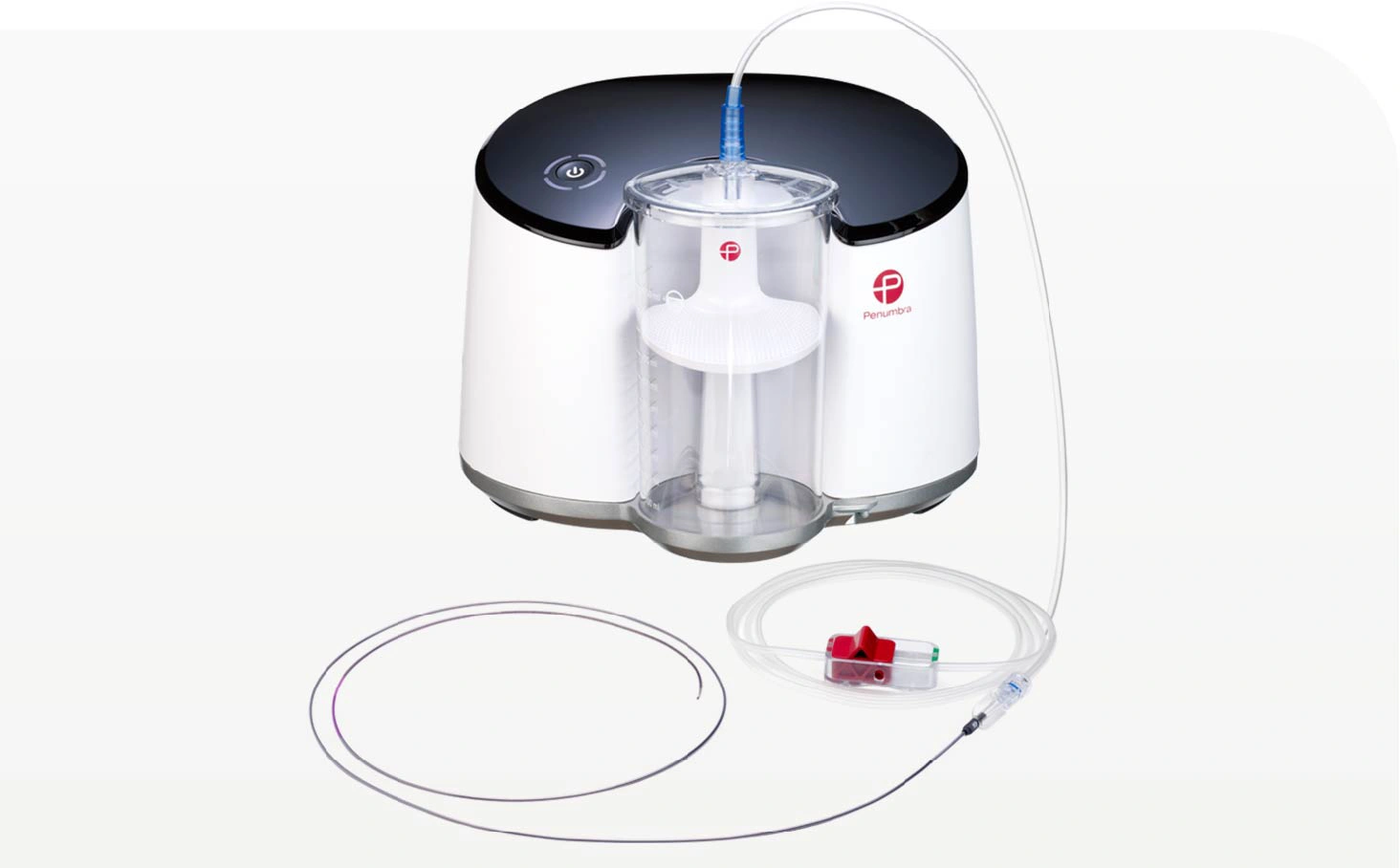
Penumbra Embolectomy Aspiration System (INDIGO™ Aspiration System)
INDIGO Aspiration Catheters and Separators: As part of the Penumbra Embolectomy Aspiration System (INDIGO™ Aspiration System), the INDIGO Aspiration Catheters and Separators are indicated for the remo
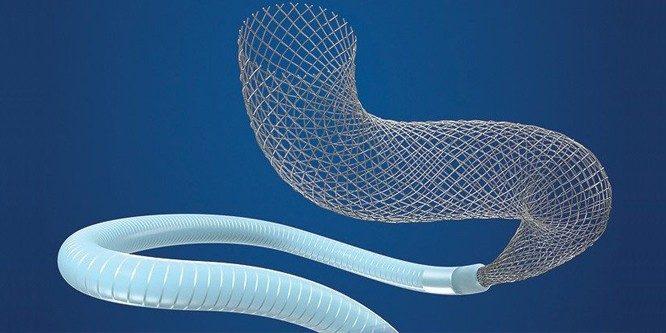
Pipeline™ Flex Embolization Device
The Pipeline™ Flex embolization device is indicated for the endovascular treatment of adults (22 years of age or older) with large or giant wide-necked intracranial aneurysms (IAs) in the internal car
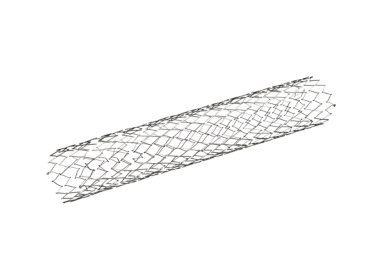
Protégé GPS Carotid Stent System
The Protégé GPS Carotid Stent System, when used in conjunction with the ev3 embolic protection systems, is indicated for the treatment of patients at high risk for adverse events from carotid endarter
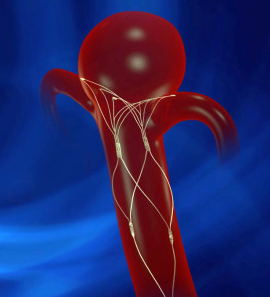
PulseRider Aneurysm Neck Reconstruction Device
PulseRider® is indicated for use with neurovascular embolic coils in patients ≥ 18 years of age for the treatment of unruptured wide-necked intracranial aneurysms with neck widths ≥ 4 mm or dome to ne

RIST Radial Access Catheter
The RIST Radial Access Catheter is indicated for the introduction of interventional devices into the peripheral, coronary and neuro vasculature. It can be used to facilitate introduction of diagnostic

SOFIATM Distal Access Catheter
The SOFIA Distal Access Catheter is intended for general intravascular use, including the neuro and peripheral vasculature. It can be used to facilitate the introduction of diagnostic or therapeutic d

ACCULINK™ Carotid Stent System
The ACCULINK™ Carotid Stent System and the RX ACCULINK™ Carotid Stent System, used in conjunction with Guidant carotid embolic protection systems, is indicated for the treatment of patients at high ri

Scepter C and XC Occlusion Balloon Catheter
It is intended for use in the blood vessels of the peripheral and neurovasculature where temporary occlusion is desired. These catheters offer a vessel selective technique of temporary vascular occlus

Target® Detachable Coils
Target Detachable Coils are indicated for the endovascular embolization of: Intracranial aneurysms; Other neurovascular abnormalities such as arteriovenous malformations and arteriovenous fistulae; Ar
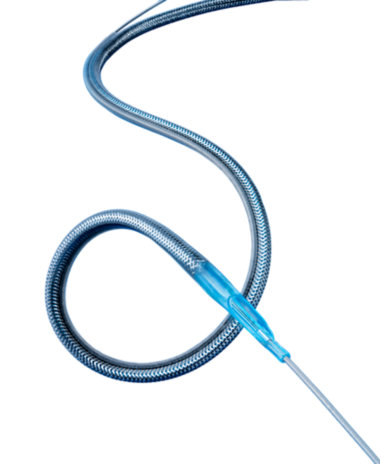
Export Aspiration Catheter
The Export Aspiration Catheter is indicated for use with the Guardwire Temporary Occlusion and Aspiration System in coronary saphenous vein bypass grafts to: 0 Contain and aspirate embolic material (

Polyvinyl Alcohol Embolic Microspheres
Polyvinyl Alcohol Embolic Microspheres are intended to be used for the embolization of arteriovenous malformations (AVMs) and hypervascular tumors, including uterine fibroids.
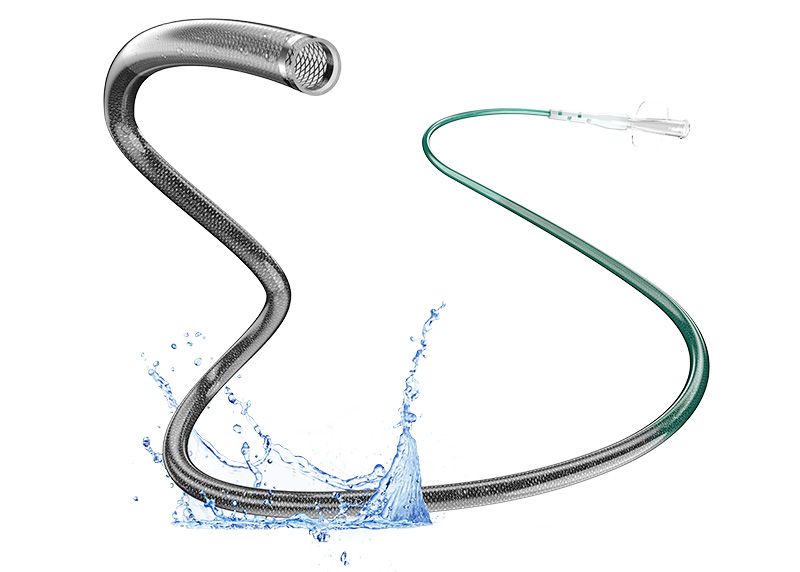
Q Distal Access Catheter
The Q Distal Access Catheter is indicated for use with compatible guide catheters in facilitating the insertion and guidance of microcatheters into a selected blood vessel in the peripheral, coronary
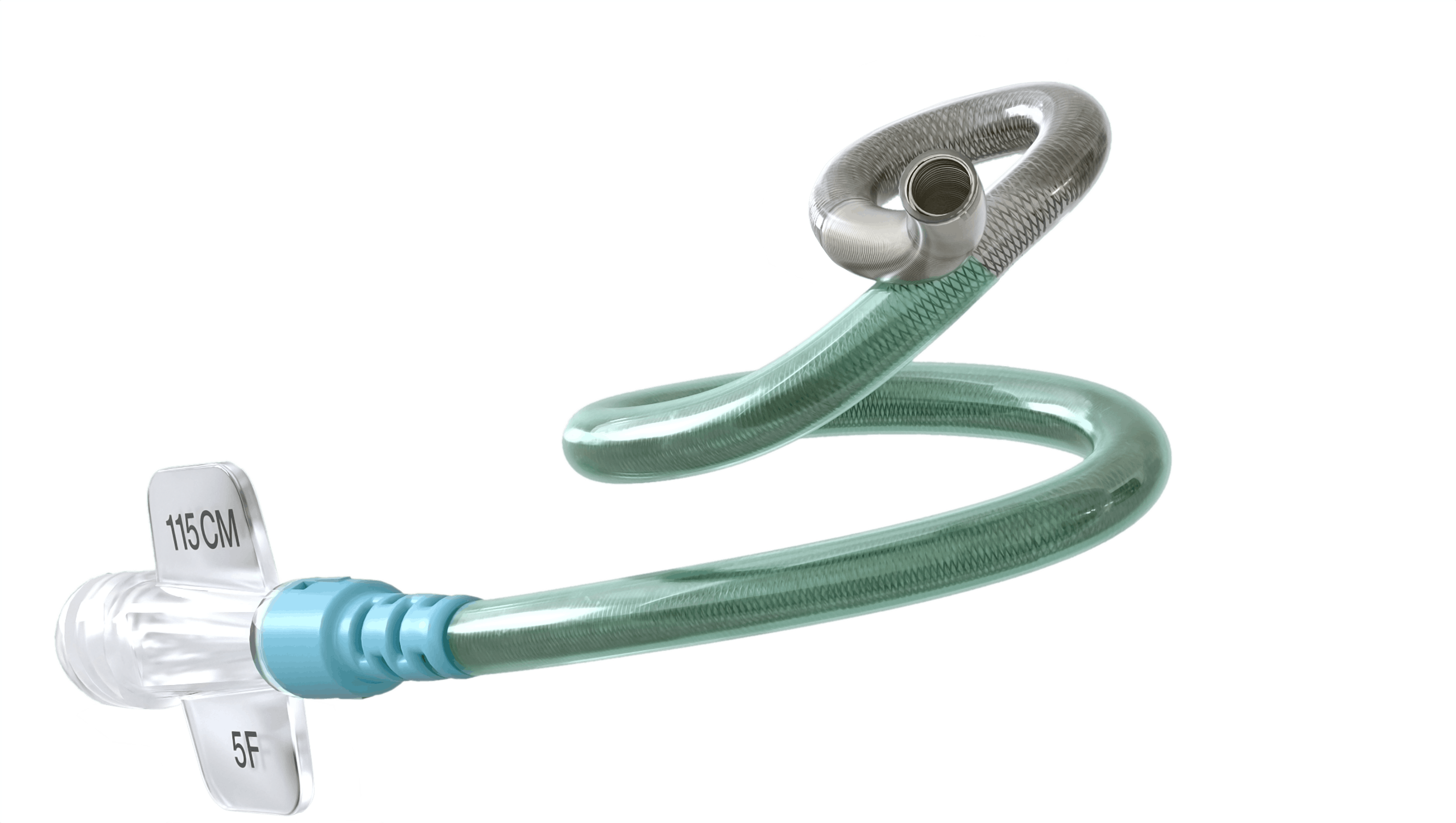
SOFIA EX Intracranial Support Catheter
The SOFIA EX Intracranial Support Catheter is indicated for general intravascular use, including the neuro and peripheral vasculature. The SOFIA EX Intracranial Support Catheter can be used to facilit

Socrates Aspiration System
As part of the Socrates Aspiration System, the Socrates 38 Aspiration Catheter with a compatible suction pump is indicated for use in the revascularization of patients with acute ischemic stroke secon

AXS Catalyst Distal Access Catheter
The AXS Catalyst Distal Access Catheter as part of the AXS Universal Aspiration System is indicated for use in the revascularization of patients with acute ischemic stroke secondary to intracranial la
Route 92 Medical Access System
The Route 92 Medical Access System is indicated for use with compatible guide catheters in facilitating the insertion and guidance of microcatheters into a selected blood vessel in the neurovascular s
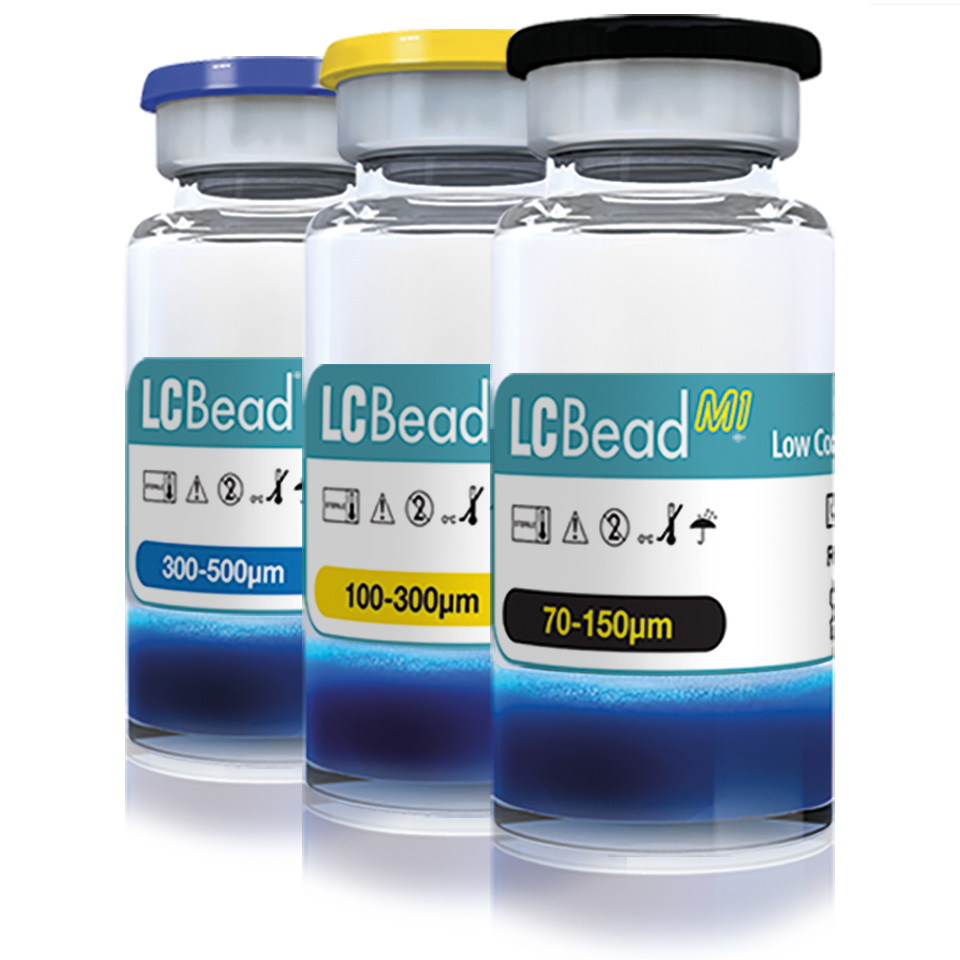
LC Bead Microspheres & BeadBlock Microspheres
LC Bead Microspheres & Bead Block Compressible Microspheres is intended for embolization of hypervascular tumors and arteriovenous malformations.

CELLO Balloon Guide Catheter
The CELLO Balloon Guide Catheter is indicated for use in facilitating the insertion and guidance of intravascular catheters into a selected blood vessel in the peripheral and neuro vasculature systems

Onyx® Liquid Embolic System
Onyx® Liquid Embolic System (Onyx®HD-500) (hereinafter called the Onyx® HD-500 System and Onyx®) is indicated for treatment of intracranial, saccular, sidewall aneurysms that present with a wide neck

Supera Peripheral Stent System
The Supera Peripheral Stent System is indicated to improve luminal diameter in the treatment of patients with symptomatic de novo or restenotic native lesions or occlusions of the superficial femoral
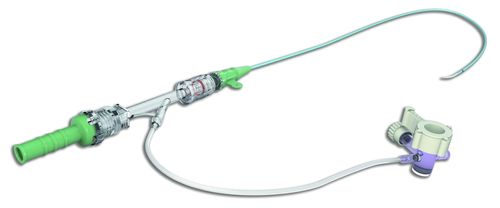
Long Sheath
The Long Sheath is indicated for the introduction of interventional devices into the peripheral, coronary, and neuro vasculature.

Xact Rapid Exchange Carotid Stent System
The Xact® Carotid Stent System (Xact®), used in conjunction with the Abbott Vascular Devices embolic protection system is indicated for the improvement of the lumen diameter of carotid arteries in pat
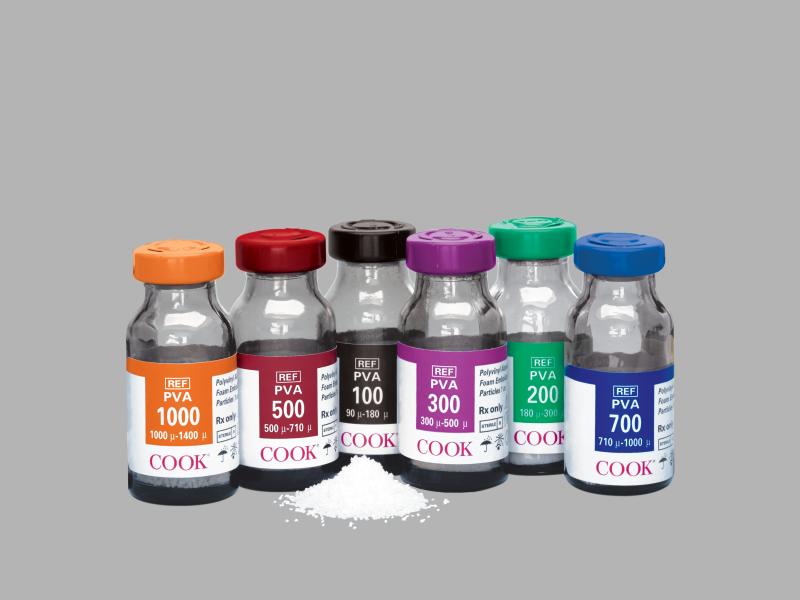
PVA Plus™ Foam Embolization Particles
PVA particles are indicated for arterial embolization of arteriovenous malformations (AVMs) and hypervascular tumors in the peripheral vasculature, and for vascular occlusion of blood vessels within t
No devices found matching your criteria.